Pioneering AGE research and technology
About us
Driving innovation in AGE research

Our mission & vision
Empowering better health decisions
We believe that early insights into biological aging can transform healthcare. Our mission is to empower professionals with reliable, science-based tools and our vision is a future where AGE measurement helps people live healthier, longer lives.
Our technology
Bridging science and daily practice
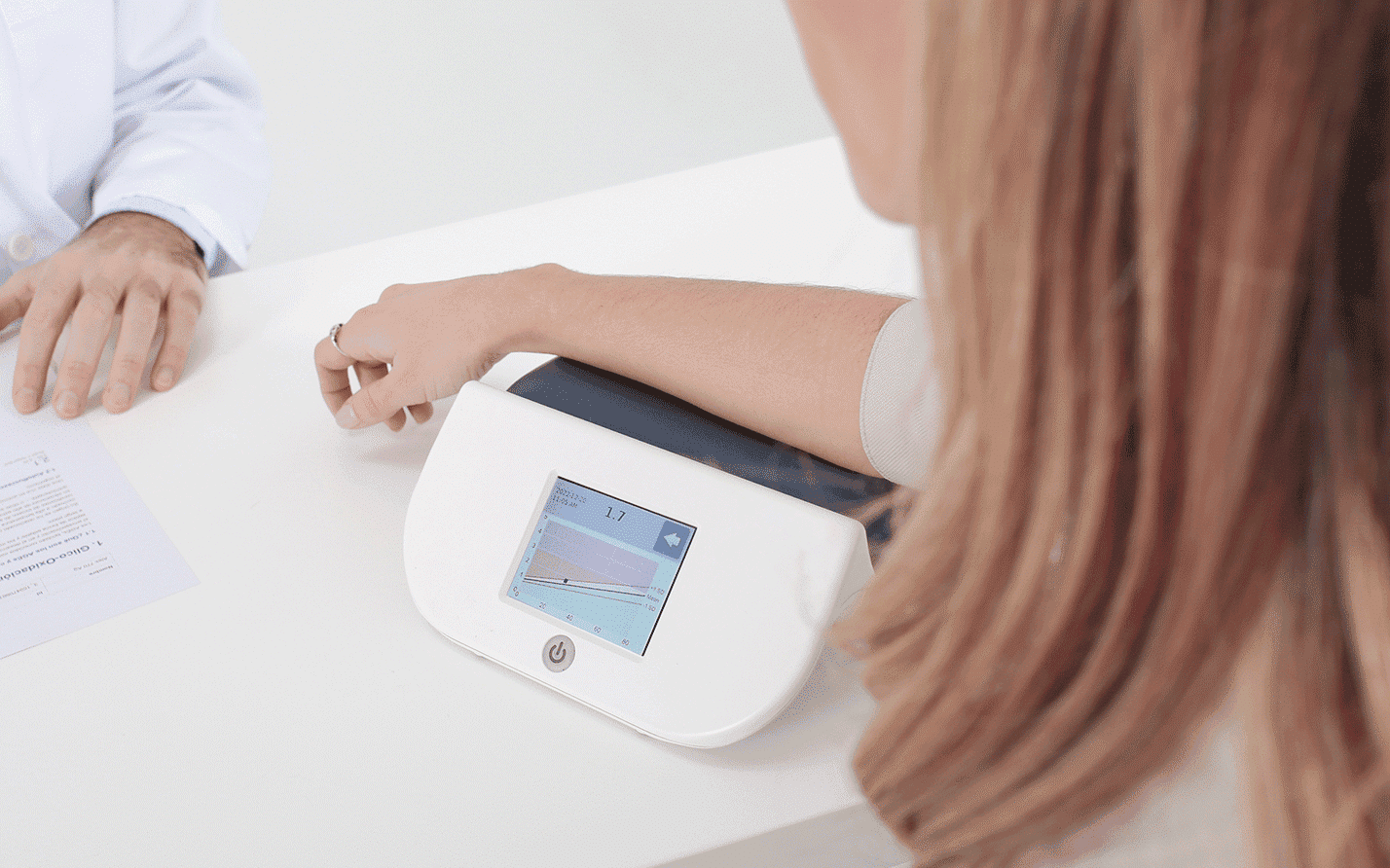
Diagnoptics developed the AGE Reader and AGE Scanner: devices that translate complex science into simple, actionable insights. Both are grounded in decades of research and validated in clinical studies.
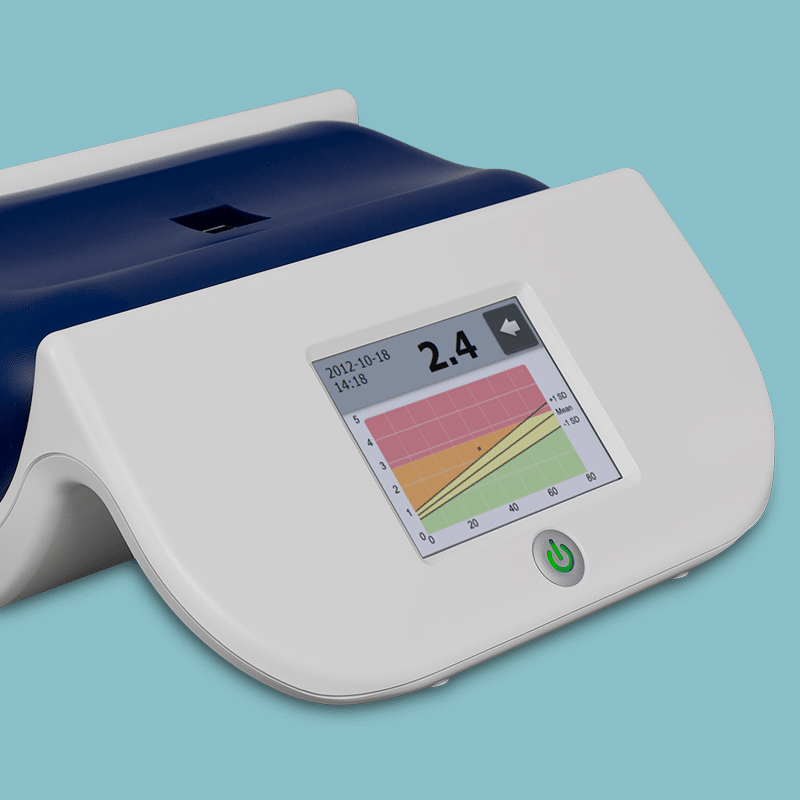
Who are we
Pioneers in AGE measurement
Diagnoptics is a spin-off from the University Medical Center Groningen, dedicated to making the invisible impact of AGEs visible. We are a multidisciplinary team of doctors, engineers, and researchers, turning science into practical solutions for health and vitality.

Science & innovation
More than 20 years of AGE research
For over two decades, Diagnoptics has led the way in AGE research and technology. Our devices are backed by hundreds of peer-reviewed studies and international collaborations.
Our history
From university lab to global leader
Founded as a spin-off from the University Medical Center Groningen, Diagnoptics has become the global leader in AGE measurement setting the standard in non-invasive health assessment.
Today
Diagnoptics continues to lead AGE measurement worldwide, combining innovation with clinical validation to advance healthy aging.
2023
Introduction of the AGE Scanner, developed for therapists and practitioners in lifestyle, nutrition, and wellness.

2020
First explorations beyond clinical settings, preparing for broader applications in lifestyle and preventive health.

2015
We believe early insights into biological aging can improve outcomes and prevent disease. Our mission is to equip professionals with reliable.

2010
AGE Reader adopted in large international cohort studies, strengthening scientific validation worldwide.
2004
Launch of the first AGE Reader, making non-invasive AGE measurement available in clinical research.
2003
Diagnoptics is founded as a spin-off from the University Medical Center Groningen, focused on AGE research and technology.

Careers
Join us in advancing AGE research
and technology
Submit an open application – we’d still love to hear from you.

Open applications
Send us your CV and motivation. We’ll keep your profile in mind for upcoming roles.
Strategic partnerships
Collaboration drives progress
We work closely with leading universities, hospitals and research institutes worldwide. These partnerships validate our technology, expand applications, and ensure that AGE measurement continues to advance clinical and practical use.

Interested in more information?
Contact us
Whether you’re interested in a product demo, research evidence or collaboration, our team is ready to assist you.